However, it is essential for manufacturers to prioritize patient safety, provide accurate information, and address any potential issues promptly. Advances in technology and increased regulatory scrutiny are expected to improve the safety and effectiveness of these devices. The Future of Spinal Cord Stimulationĭespite the legal challenges faced by Abbott and other manufacturers, spinal cord stimulation remains a viable treatment option for chronic pain management. It is crucial for these victims to seek justice and hold the manufacturer accountable for their suffering. Many individuals have experienced worsened pain, additional medical complications, and emotional distress due to the alleged defects in Abbott’s products. The Abbott spinal cord stimulator lawsuit has had a profound impact on patients who trusted the device to alleviate their chronic pain. The amount of compensation awarded depends on factors such as the severity of injuries, the impact on the victim’s life, and the strength of the evidence presented during the lawsuit. Victims of defective medical devices may be eligible to receive compensation for various damages, including medical expenses, pain and suffering, lost wages, emotional distress, and future medical needs. These lawyers can guide you through the complex legal process, help gather evidence, and build a strong case against Abbott to increase your chances of receiving fair compensation for your suffering. 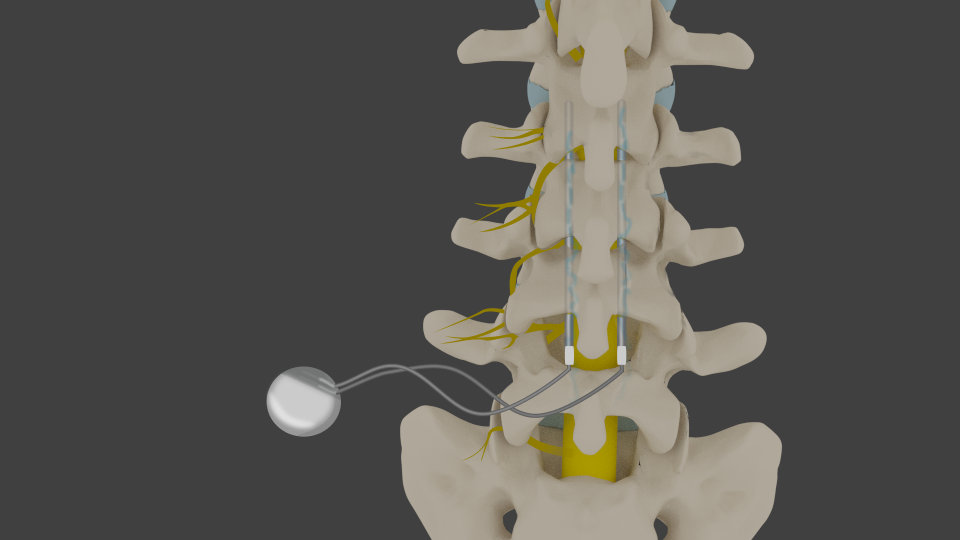
If you or a loved one have been affected by the Abbott spinal cord stimulator, it’s crucial to seek legal representation from experienced attorneys specializing in medical device litigation. In the case of the Abbott spinal cord stimulator lawsuit, plaintiffs are taking legal action against the company to hold them accountable for their alleged negligence. The process typically involves gathering evidence, filing a complaint, discovery, negotiations, and, if necessary, going to trial. When individuals believe they have been harmed by a medical device, they have the right to seek compensation through a lawsuit. Some patients have experienced device failure, migration of the leads, infections, or ineffective pain relief, leading to further medical complications and emotional distress. 
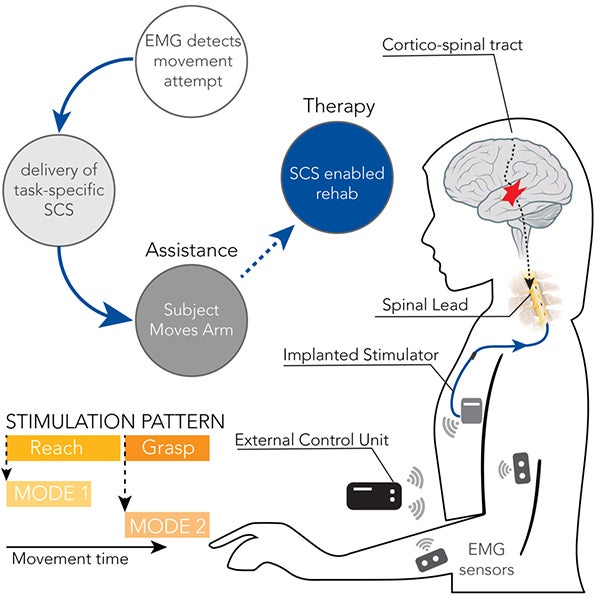
Allegations against Abbott include claims of faulty devices, inadequate warnings, and misleading marketing practices. In recent years, several patients who received Abbott spinal cord stimulators have reported adverse effects and complications. These devices consist of a battery-powered pulse generator, placed under the skin, and thin wires with electrodes that are inserted into the epidural space. Spinal cord stimulators (SCS) are implantable devices that deliver electrical pulses to the spinal cord, thereby interfering with pain signals and providing relief to individuals suffering from chronic pain conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
